Sepsis is horrible.
When the immune system goes into overdrive during an infection, little can be done. Sepsis kills millions each year, and treatments are limited.
Can we find hope in coated viruses delivering RNA editors?
Today’s paper believes so.
Don’t keep this newsletter a secret: forward it to a friend today!
Was this email forwarded to you? Subscribe here!
🧬 Some personal news! Tomorrow (February 27th) is my last day as a postdoc.
What am I going to do now? I’m not sure. For now, I’ll focus on writing and resting.
But you can look forward to the usual weekly dose of science! And I have a few new projects in mind. The “rest” part might be optimistic…
For the moment, let’s get into today’s paper.
Viruses Get Armor
Scientists developed innovative biomineralized lentiviruses that deliver RNA editors to treat sepsis, reducing inflammation and organ damage. Image credits: Nature.
Sepsis: Immune System on Overdrive
Sepsis is brutal.
It kills millions every year. Instead of fighting infections, immune cells spiral into overdrive, releasing massive amounts of inflammatory molecules that damage tissues and organs.
In short, sepsis is the most dangerous immune system failure.
Despite decades of research, treatments are limited. Doctors try to fight the infection, but controlling the immune system is incredibly difficult. In 2017 alone, sepsis killed 11 million people.
At the center of this storm are macrophages.
Normally, they’re the key to regulating inflammation. But during sepsis, the excessive activation of M1 macrophages leads to the uncontrolled release of pro-inflammatory cytokines and organ failure.
In all of this, the NLRP3 inflammasome plays an important role.
Inflammasomes are cytosolic multi-protein complexes that sense microbial molecules and activate inflammatory defences. But when these responses run wild, you get cytokine storms, cell death, and organ collapse.
So, could NLRP3 be the target we're looking for?
RNA Editing to The Rescue
RNA editing is a powerful new solution.
Instead of rewriting DNA, you modulate protein expression by targeting RNA. It’s based on the CRISPR-Cas13 system, naturally capable of targeting and cutting RNA.
Over the years, researchers have found proteins with new functions or improved efficiency. CasRx is a great example: it’s efficient, specific, and has low off-targets. And it doesn’t touch the DNA: no permanent changes!
Problem solved then? Well, it’s a start. CasRx does offer a safer alternative to other, more genotoxic or less efficient methods, but it still has a big, familiar problem.
Delivery is a nightmare.
You need to get these molecular machines into the right cells, in the right place, at the right time. In vivo, you have a few options:
Viral vectors: Adeno-associated viruses, lentiviruses, virus-like particles. Efficient, but immunogenic and persistent.
Lipid nanoparticles: Non-viral, they offer reduced immunogenicity, and are great for liver targeting, but not much else.
Other non-viral methods: Polymers, DNA origami, engineered proteins: still niche.
Viral vectors are, hands down, the most common delivery method. They’re powerful, but have drawbacks:
Low delivery efficiency: Reaching enough cells to have a therapeutic effect is challenging.
Immunogenicity: The human immune system reacts to the viral proteins, especially with repeated injections.
This limits systems like CasRx. Did we reach another dead end?
Giving Viruses an Armor
Not according to the authors of today’s paper.
The team developed a macrophage-targeted, chemogenetically controlled RNA editing therapy for sepsis. The central idea is to degrade NLRP3 mRNA in macrophages to reduce immune system overactivation.
They combined RNA editing, targeted delivery, and temporal control to treat sepsis.
And it worked: in mouse models of sepsis, it reduced NLRP3 mRNA and pro-inflammatory cytokine levels, protected organs, and improved survival. Let’s look at the details!
Building the Platform
The system has a few parts. Let’s nail them down.
Destabilized CasRx (dsCasRx)
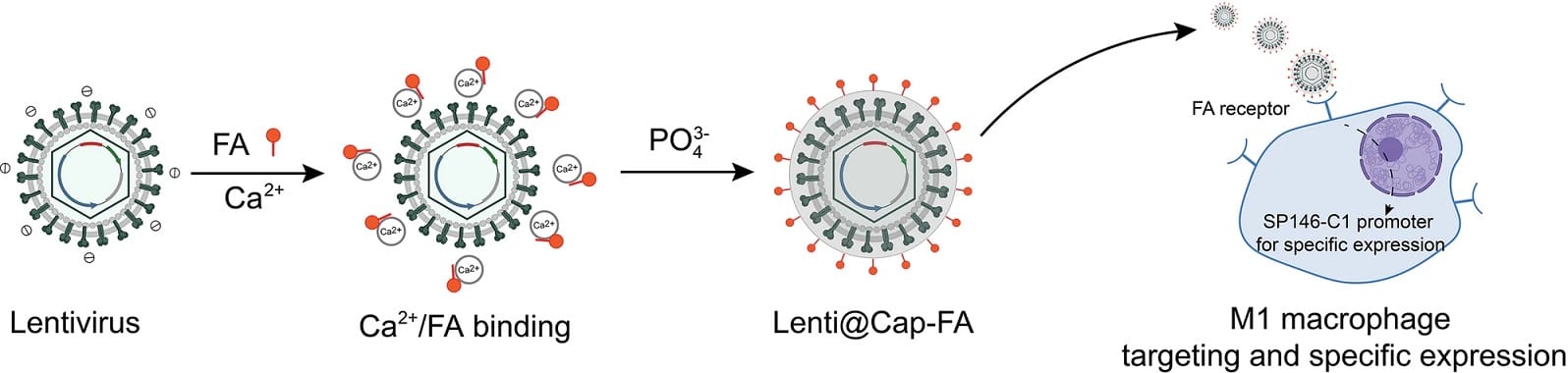
The team fused CasRx to a destabilizing bacterial domain, so it’s rapidly degraded. That is, unless it’s stabilized by trimethoprim (TMP). This enables temporal control: no TMP → CasRx degraded; TMP present → active editor. Plus, TMP is an antibiotic, so you get a positive, synergistic effect on the infection. The target of CasRx is the NLRP3 mRNA.Macrophage specificity
To ensure expression only in macrophages, CasRx is under the control of a macrophage-specific promoter.Biomineralized lentivirus (Lenti@Caps)
My favourite part! The team coated the lentiviruses with a calcium phosphate shell they called “Cap”, creating Lenti@Cap. This shell protects viral proteins, reduces immunogenicity, and allows for repeated administration. A suit of armor for the viruses!
Folate (FA) functionalization (Lenti@Caps-FA)
FA binds to the folate receptor-β, overexpressed on inflammatory M1 macrophages. So, they added FA to the biomineralized viruses to form Lenti@Cap-FA.
TMP encapsulation
TMP (the CasRx stabilization trigger) is encapsulated in polymeric micelles to improve solubility and delivery.
Lots of innovations!
This is how it works in a nutshell:
Inject Lenti@Cap-FA carrying the dsCasRx/sgRNA DNA.
The viruses enter the macrophage, the Cap dissolves in endosomes, and the construct integrates into the genome.
The macrophage-specific promoter expresses dsCasRx.
If TMP is present, dsCasRx cuts NLRP3 mRNA and inhibits inflammasome formation.
The macrophage produces fewer pro-inflammatory molecules, reducing organ damage!
Now, that sounds really cool. But you know the real question.
Does it work?
Fighting Sepsis in Vivo
The team first tested the system in vitro.
Everything works. TMP stabilizes dsCasRx, leading to NLRP3 knockdown. Increasing TMP doses reduces NLRP3 more. Removing TMP reverses the knockdown, and re-adding reactives the editing.
A tunable and repeatable system! Awesome. Does it work in vivo?
The team tested it in a well-used mouse model. They performed cecal ligation and puncture to induce sepsis (yeah, not great). Then, they injected Lenti@Cap-FA encoding dsCasRx and encapsulated TMP.
The Lenti@Cap-FA localized to the infection site, where M1 macrophages accumulated, and FA targeting increased macrophage uptake vs untargeted particles.
In addition, the lentiviral shell enabled up to five repeated administrations without significant side effects! You can’t do this with uncoated lentiviruses, because pre-existing antibodies rapidly remove them.
The treatment also had positive therapeutic effects:
Survival: It increased survival to 80% at 8 days versus 0% in PBS control. This comes from the synergistic activity of TMP killing the bacteria and Lenti@Cap-FA calming the immune system.
Clinical scores and body weight: Treated mice showed improved both physical and mental clinical scores. Plus, after an initial weight loss because of the sepsis, they put it back on!
NLRP3 and cytokines: Peritoneal macrophages from treated mice showed reduced NLRP3 mRNA and protein, and lower pro-inflammatory cytokines (TNF-α/IL-1β/IL-6).
And finally, if the mice had a secondary infection, simply reactivating the system with TMP preserved survival: on-demand reactivation potential!
A New Weapon Against Sepsis?
A super cool paper! Well written, and I’ve learnt a lot.
I knew nothing about sepsis. Now I know it’s horrible. The authors combined 3 different, complementary innovations to create a powerful system:
Biomineralized lentiviral camouflage
Chemogenetic control
Dual macrophage specificity.
Together, they create a modular platform for tunable, cell-type-specific RNA editing in vivo. And the mouse data show clear therapeutic benefits!
Now, it’s not perfect, and the authors know it:
Genome integration risks: Lentiviral integration can cause long-term problems; integration-deficient viruses or a completely different delivery method could be solutions.
Administration route and targeting: The system is less suitable for systemic delivery. Also, FA is good, but it’s not that specific for targeting: it would be great to use better systems.
Immunogenicity: Biomineralization improves it, but it doesn’t eliminate antibody responses on repeated dosing. We need better ways to avoid immune recognition!
But all in all, an incredible approach! I hope it moves further, because sepsis looks brutal, and the lack of treatments is saddening.
Also, a word on the mouse models. I get that they’re practically mandatory for immunology work. But I still don’t like them, and it would be great to see more work done on in vitro models.
But for the moment, go here and read this paper! It’s a cool one.
If you made it this far, thank you! What do you think of RNA editing? Do you think it has a place in biomedicine? Reply and let me know!
P.S: Know someone interested in RNA editing? Share it with them!
What did you think of today's newsletter?
More Room:
Rotating Amino Acids to the Rescue: To study proteins, you generally need large fluorescent tags. These can be a problem for the protein function and limit spatial resolution. In this work, the authors develop a strategy to site-specifically incorporate environment-sensitive, rotor-based noncanonical amino acids into proteins in mammalian cells. By engineering aminoacyl-tRNA synthetases, they enable precise labeling of individual residues, allowing real-time detection of local microenvironment changes during processes such as protein aggregation and clustering. This approach provides high-resolution insight into protein dynamics without large fluorescent fusions.
β-Barrels Go Places: In science, some themes are popular, and others are not. For example, β-barrel nanopores are key in biology and are used for sequencing, but how they work is still unclear. This study identifies two key behaviors in β-barrel nanopores: open-pore rectification and gating. Rectification stems from charge-driven ion accumulation in the pore, while gating results from electric-field-induced counterion dissociation that triggers structural changes. The authors used the findings to optimize aerolysin for nanofluidic computing.
Is Phage Medicine Coming? Phage therapy is very promising, but it’s dependent on the availability of phages to attack bacteria. Sometimes, that number is low, like in the case of mycobacteria. So, in this study, the authors develop a method for de novo synthesis and assembly of high–G+C mycobacteriophage genomes, overcoming challenges in DNA synthesis and genome size. Using enzymatic DNA synthesis and High-Complexity Golden Gate Assembly, they reconstruct complete phage genomes and reboot them in bacteria, generating engineered variants with targeted mutations or added genetic payloads. Incredible work!
