Vaccines saved six lives every minute between 1974 and 2024! Insane.
mRNA vaccines are the new kid on the block, and they helped with the COVID-19 pandemic, after decades of R&D. Can we combine them with computationally designed proteins to make them even more powerful?
Read on to find out!
Don’t keep this newsletter a secret: Forward it to a friend today!
Was this email forwarded to you? Subscribe here!
mRNA Builds Vaccines
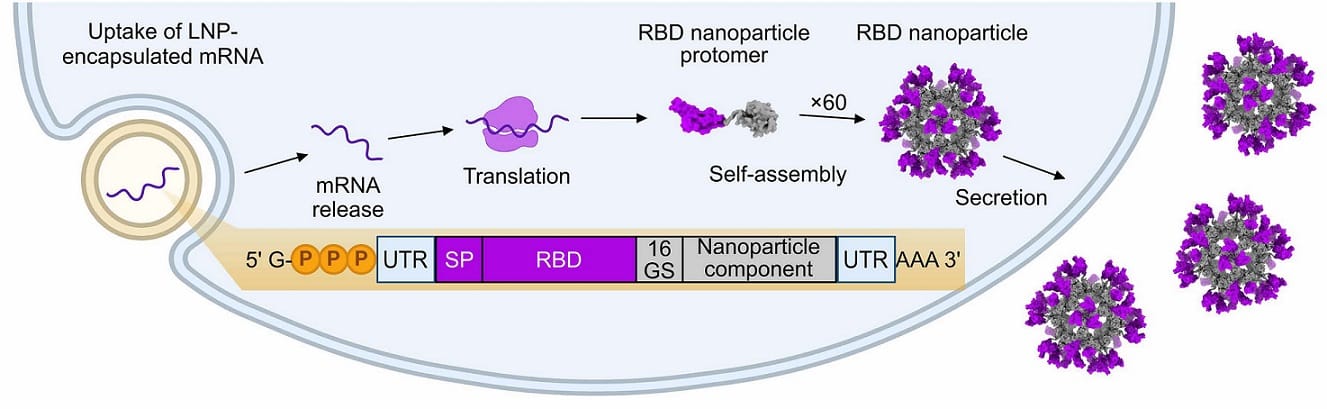
Researchers developed “mRNA-launched” vaccines that combine the most powerful features of mRNA and protein vaccines to enhance immune response. Image credits: Science.
Vaccines: The Most Powerful Weapons against Diseases
Are vaccines the greatest healthcare invention ever?
Probably. Over the past 50 years, vaccines have saved an estimated 154 million lives, mostly children under 5. This means six lives saved every minute! Pretty insane. The big ones were the vaccinations against measles, polio, and tuberculosis.
And never forget that vaccines eradicated smallpox!
Vaccines are our most powerful weapons against diseases. We’ve all seen their power during the COVID-19 pandemic. The progress in vaccine technology has been incredible!
There isn’t only one way to build a vaccine. There are six main types, each with strengths and trade-offs:
Live-attenuated vaccines: A weakened form of the pathogen that can still replicate. They produce strong, long-lasting immunity.
Inactivated vaccines: A dead version of the pathogen. Safer for immunocompromised people, but often needs boosters.
Toxoid vaccines: Inactivated bacterial toxins teach the immune system to fight that rather than the germ itself.
Viral vector vaccines: A safe version of a different virus delivers genetic instructions into cells (for example, DNA for viral proteins).
Subunit vaccines: Pieces of the pathogen (proteins or sugars) provide a focused immune response.
mRNA vaccines: Directly instruct cells to create a protein component of the virus, triggering an immune response. Often use lipid nanoparticles (LNPs) for delivery and to increase immune system activation (adjuvant effect).
Plenty of options to choose from!
mRNA + Proteins: The Best of Both Worlds
Can we mix these modalities to improve vaccines?
Exactly what the authors of today’s paper had in mind!
They focused on mRNA vaccines and protein vaccines. In particular, protein nanoparticle vaccines that display multiple copies of an antigen (multivalent display). Up to 60!
They each bring their own immunological advantages:
mRNA: Intracellular antigen expression, robust CD8 T cell activation, and rapid manufacturing.
Protein nanoparticles: Strong B cell activation and neutralizing antibody responses.
Perfectly complementary!
So, the team designed an mRNA sequence that encodes a multivalent, self-assembling nanoparticle, secreted and assembled by host cells!
They dubbed this strategy “mRNA-launched” vaccines. Their goal is to speed up vaccine development for new pathogens. And what better test than SARS-CoV-2?
Sounds awesome, right? Let’s see how they did it!
Designing and Engineering: What They Built
The designed mRNA-launched vaccine has 3 main components
The scaffold: I3-01NS scaffold
A computationally designed, one-component 60-subunit icosahedral particle previously optimized for secretion from mammalian cells. The mRNA enters the cell → protein monomers are made → they self-assemble into nanoparticles.
The antigen: RBD or Rpk9?
The plan was to fuse the standard receptor-binding domain (RBD) of SARS-CoV-2 (the Wuhan strain). That didn’t work; the cells didn’t secrete any particles. So, they switched to Rpk9, a stabilized RBD variant with 3 mutations that increase expression and secretion.
The mRNA construct
The team fused Rpk9 to the N terminus of I3-01NS with a flexible linker. A signal peptide and a secretion element complete the mRNA design. Finally, they packaged the nucleoside-modified mRNA into LNPs for delivery!
And voilà! A genetic blueprint for a self-assembling vaccine.
The team tested it in vitro by transfecting mammalian cells. RBD-I3-01NS didn’t secrete, but Rpk9-I3-01NS did! At around ~29.9 mg/L, which seems to be a reasonable yield (with non-assembling Rpk9 versions secreted at ~122 mg/L, for reference!).
The purified nanoparticles showed the right size and icosahedral shape in DLS and negative stain electron microscopy. And they also bind to antibodies, confirming that the antigens are intact and accessible on the assembled nanoparticles!
Time to check if it actually works!
Comparing Vaccines in Mice
The team compared lots of vaccine modalities and constructs in mice, across different experiments:
mRNA-launched Rpk9–I3-01NS (secreted 60-mer nanoparticle→their system)
standard mRNA delivering membrane-anchored spike (S-2P; exact sequence used for real vaccination!)
mRNA delivering secreted Rpk9–I53-50A (nonassembling trimer scaffold)
Protein vaccines: purified Rpk9–I3-01NS nanoparticle, Rpk9–I53-50 trimers, S-2P protein controls
Controls: luciferase mRNA, empty LNP, PBS
Generally, the mice received a first (prime) dose at week 0 and a boost at week 3.
Immune Readouts: Antibodies and T Cells
First, the team checked the antibody response.
At all tested mRNA doses, mRNA-launched Rpk9 nanoparticles induced antigen-specific binding titers:
4x higher than standard mRNA vaccines!
10x higher than secreted non-assembling Rpk9
In all cases, the mRNA-launched multivalent display was better than using single proteins!
After the boost, mRNA-launched Rpk9 nanoparticles induced:
5-fold higher titer of neutralizing antibodies than traditional mRNA vaccines, at every dose
7-fold higher neutralization at the 5-µg mRNA dose!
And a tiny 0.2 µg dose of mRNA-launched Rpk9–I3-01NS elicited neutralizing titers comparable to 5 µg of traditional mRNA vaccine!
All this, while maintaining the strong CD8 T cell response you get with mRNA vaccines!
Protection Results
The real test of a vaccine: can it protect from pathogens?
Protection from SARS-CoV-1 Wuhan-Hu-1
They immunized the mice once with different vaccines or controls, and 5 weeks later, the mice were infected.
The results were great. The group treated with mRNA-launched had no weight loss, minimal lung damage, and the virus was undetectable in the lung. The other groups showed weight loss up to 20%, lung damage, and detectable virus load.Challenge with Omicron BA.5 variant
The infamous Omicron! Do you remember it? The team immunized the mice with a prime-boost regimen (0 and 4 weeks), then infected them at week 9 with the virus.
The mRNA-launched vaccine protected the mice against weight loss and lung disease, showing protection against different variants!
A New Age for Vaccines?
Awesome article!
This new “mRNA-launched” approach is cool and powerful:
It goes head-to-head with clinically approved vaccines
It outperforms them! Combining the strengths of mRNA and protein vaccines
The platform is modular: the authors applied it to other sarbecoviruses (the SARS-CoV family) and to HIV (with eOD-GT8, do you remember that?)
The authors also noted some limitations:
In vivo assembly and secretion are not directly measured.
Some antigen/scaffold combinations just don’t work!
Mouse → human translation is unknown. We won’t know until clinical trials. I’m not a big fan of animal testing, you know it.
Plus, biodistribution, durability, and mechanism still have to be properly studied.
But these “mRNA-launched“ computationally designed protein nanoparticle vaccines are cool! They combine the T-cell activation of mRNA vaccines with the B-cell-activating potency of multivalent display.
With the manufacturing speed of mRNA vaccines! That would be awesome.
So, cool article: read all of it here!
If you made it this far, thank you! What do you think of mRNA vaccines? Do you think they have a place in biomedicine? Reply and let me know!
P.S: Know someone interested in SynBio? Share it with them!
What did you think of today's newsletter?
More Room:
Protein-Based… batteries? I love the ideas of bio-based electronics, I’m not gonna lie. And we’re slowly getting there! In this study, researchers engineered self-assembling protein biomaterials with added glutamic acids to create charge pathways that enhance ionic conductivity. The modified proteins form films with ~10× higher conductivity, further improved by salt addition, due to enhanced proton transport and ion diffusion. These films can function as electrolytes and separators in supercapacitors, showing potential for sustainable energy storage and bioelectronics.
Biomechanics Goes In Vivo: Mechanical stresses are important in living organisms. Development, disease, and more! Putting these insights into use, though? Hard. until now! In this study, the authors develop a method to enhance tissue regeneration by covalently linking cell integrins to a hydrogel using bioorthogonal click chemistry. This stable integrin–hydrogel connection ensures that mechanical stress is consistently transmitted to the cell nucleus, activating regenerative responses in transplanted cells regardless of environmental conditions.
Rolling Nanoclusters into CRISPR: Gene editing is full of potential, but it also still has many problems. One? Escaping from lysosomes after entering a cell. But maybe this study brings a solution! Researchers develop magnetically driven nanoparticle “nanorobots” to improve intracellular delivery of CRISPR–Cas9. These ~200 nm magnetic clusters can be propelled by external magnetic fields, helping them move through viscous cytoplasm and escape lysosomes. This boosts gene-editing efficiency and enables editing of targets such as PD1 and PLK1, even in difficult-to-transfect cells, offering a promising platform for gene therapy.
